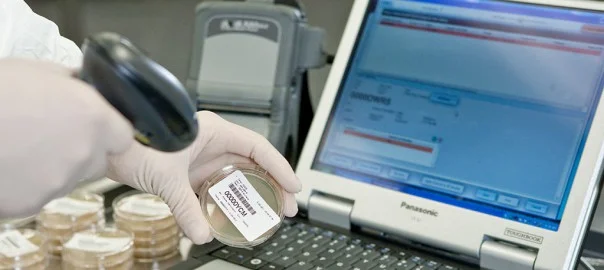
MODA-EM®
Eliminate unnecessary time and cost associated with paper-based QC methods
The MODA® Platform delivers a comprehensive informatics platform that automates quality control (QC) processes for all regulated manufacturing in the Life Sciences industry. As a user of The MODA® Solution, Lonza has direct experience with its value – providing improved decision making, regulatory compliance, and productivity across our own global manufacturing facilities
By combining automated scheduling, workFLows, mobile data acquisition, device integration, and advanced analytics, The MODA® Platform delivers efficiencies across your organization.
QC Departments Are Able To:- Automate data collection from devices and people -Eliminate redundant data entry and transcription errors -Gain direct traceability of QC Micro programs -Increase worker efficiency -Improve regulatory compliance -Make sound product quality and release decisions -Quickly advance Green initiatives
Measurable Benefits:- QC Technicians quickly and accurately collect data in critical areas – Lab Managers improve process efficiency for scheduling and tracking. – Supervisors gain immediate, detailed reporting on the production area to enable sound product release decisions, effective investigations, and quick response to audits. – Executive Management can access longer term trend reports to assess overall program effectiveness.
The MODA® Platform
The MODA® Platform is a comprehensive solution that bridges the gap that currently exists between manufacturing and QC to provide a single batch record with an intuitive review and approval interface. With GMP compliance and tracking at its core, The MODA® Platform combines data capture and error prevention with the flexibility to capture and trend key quality and performance metrics, both in the lab and on the production floor.
VHPS Pass Box
When it comes to decontamination of the outer surface of the materials, we have an excellent solution which follows the Dry VHP technique. Steri Transfer is designed for the materials transfer from Low grade area to Critical area(Class A/B area) at Low temperature. Passing pharmaceutical raw materials, tools and other materials into Class B district.
ISOLATOR
Hangzhou Tailin Bioengineering Equipments Co., Ltd. has been committed to the development of Sterility isolator technology. It can use in inbuilt sterility test pump and built in VHP generator. The effectiveness of Bio-decontamination achieves lg6 level. With options of flexible wall or rigid wall isolators it is an isolator which is technically one of the best.
Microbial Limit Tester & Filteration Funnel
This Product is designed and manufactured for the microbial test, it has integrated diaphragm pump which could directly drain. So, it doesn’t need filtering flask.
Quantek Head Space Oxygen Analyser
Quantek is an expert for analysing various gases especially Oxygen in Vials. Model 905V is one of the most successful and robust models in the Industry. Pharmaceutical preparations packaged in vials are often gas-flushed to provide a specific atmosphere in the vial, with defined levels of oxygen, nitrogen or other gases. Depending on the product, these vials can range in size from 1 ml to 250 ml or larger. Small vials present some difficulty for oxygen analysis because the sample volume available is often quite small. A one ml vial, for example, may have only 0.5 ml of headspace gas. For direct analysis with a pharmaceutical oxygen analyzer, the instrument must be capable of analyzing very small volumes. Our pharmaceutical oxygen analyzer is designed for this process.
METONE 3400+
The MET ONE 3400+ helps you manage accurate cleanroom monitoring data via safe and secure internal software and web access-there’s no external software needed and All data is encrypted
It uses Microsoft Active Directory for Username and Password Control for log-on and electronic signatures, it supports 21 CFR Part 11 compliance and ALCOA guidance
MET ONE SIMPLY PAPERLESS
MET ONE Simply Paperless automatically exports data direct to a USB memory stick, straight through your Ethernet or routed via Wi-Fi network. No software required.
CONTINUOUS/ONLINE AIR PARTICLE COUNTER -INBUILT PUMP
MET ONE 6000P series remote airborne particle counter is the most reliable top performing, high sensitivity remote particle counter with a built-in pump. Met One 6000P series is designed to meet the specific needs of cleanroom operations within the pharmaceutical, semiconductor, hard disk drive and flat panel display industries. With an array of industry leading features and simplified installation options, the MET ONE 6000P is easy to integrate with any facility monitoring system.
CONTINOUS/ONLINE AIR PARTICLE COUNTER-EXTERNAL PUMP
The Met One 6000 series remote airborne particle counter offers accurate and reliable continuous particle monitoring. With an array of communication and mechanical installation options, the Met One 6000 is easy to integrate with any facility monitoring system. These features also reduce downtime for instrument removal and reinstallation during routine calibration and preventative maintenance cycles. Built with Long Life Laser™ technology, the MET ONE 6000 offers industry leading diagnostic features that reduce troubleshooting time and related downtime costs.
Let's connect and discuss further
Tell us about your requirement and we will get back to you soon
Need Assistance?
 +91 98245 10383
+91 98245 10383 sales@shreedhargroup.com
sales@shreedhargroup.com